Cyril Tordeur , Université Libre de Bruxelles (ULB ) Consider, on the one hand, an 80-year-old patient, out of breath with the slightest effort, whose weakened heart is allowing blood to flow back to the lungs, and, on the other, a trained astronaut aged 35 to 45, who has just returned from six months aboard the ISS, the International Space Station, and who appears to be in perfect health. What could these two individuals possibly have in common?
For the first time, our work has demonstrated the existence in humans of a phenomenon previously observed only in animals: in space, certain small muscles housed within the heart chambers atrophy, despite the sport that astronauts do every day.
In reality, the astronaut’s mission has left its mark on his heart. Behind his apparent physical shape lie subtle cardiac changes that mimic, in accelerated form, what aging has caused much more slowly in the terrestrial patient. The culprit is weightlessness, a state in which the body’s own gravity is no longer felt, causing those who venture into space to float. A situation that could complicate future interplanetary voyages.
The astronaut’s heart in weightlessness
Selected from thousands of applicants, astronauts are professionals who undergo rigorous training and continuous medical monitoring. However, as soon as they leave Earth, their cardiovascular system undergoes a process of "deconditioning". In other words, their physiology and physical characteristics change, because they simply don’t need to work as hard any more.In weightlessness, the heart no longer has to struggle so hard to pump blood to the brain. Body fluids are redistributed to the head, blood volume decreases, and the heart muscle, less solicited, adapts.
Scientists, who have been studying these phenomena since the first space missions, have discovered that this adaptation is not without consequences. Earlier studies had shown, for example, that the heart could lose muscle mass after a few weeks in orbit, and that its shape became more spherical.
Over thirty years ago, a study carried out on rats also revealed a reduction in the size of small, very specific cardiac muscles - the papillary muscles.
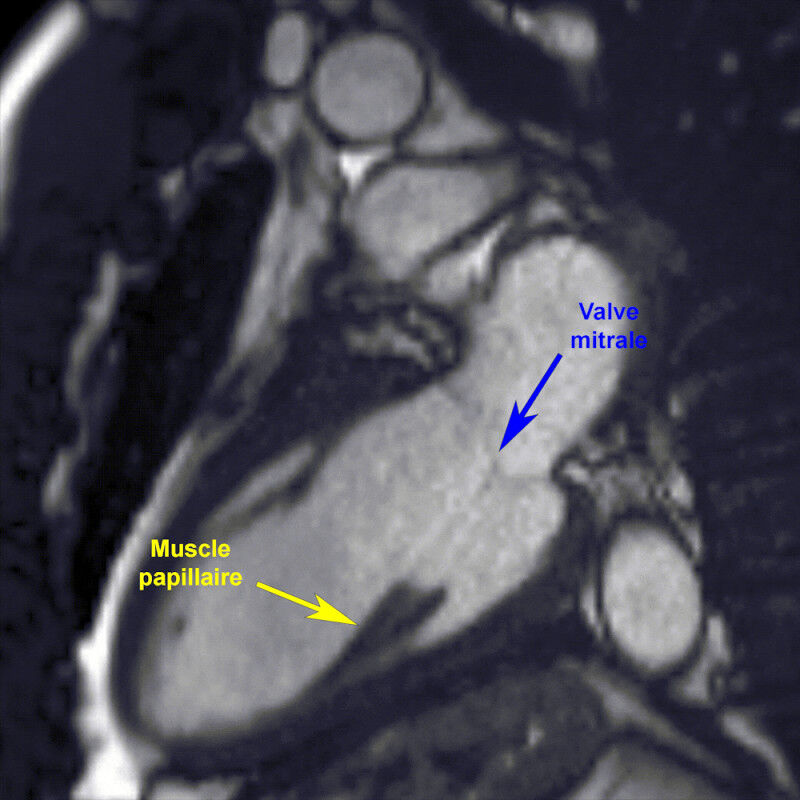
Papillary muscles, essential but little studied
The heart’s main mission is to circulate blood through the lungs and the body. To do this, it is made up of four chambers that contract at a regular rate. Valves act as non-return valves, preventing blood from flowing in the wrong direction.To perform their function, they are aided by small muscular structures that make up less than 10% of the heart’s total mass: the papillary muscles. These prevent the valves from flipping over like an umbrella in strong winds when the heart contracts. In fact, this is precisely what happens in the case of mitral insufficiency, a condition that affects millions of - mainly elderly - people worldwide.
In 1992, a study carried out on rats showed a reduction in the size of these papillary muscles after just two weeks in space. However, no such study had been carried out on humans during long-duration space flights.
To fill this gap, we used magnetic resonance imaging (MRI) to precisely measure the mass of these small muscles in astronauts assigned to long-duration space missions (six to twelve months) aboard the ISS. The measurements were taken between forty-five and sixty days before lift-off and around a week after their return to Earth.
A risk of mitral valve leakage
Our results showed an average 14% reduction in papillary muscle mass after space flight. This selective atrophy, combined with the increased sphericity of the heart observed in weightlessness and a 6% increase in the diameter of the mitral valve (located between the left atrium and the left ventricle), creates anatomical conditions that could theoretically favor a lack of seal of the said valve.In such a case, blood is no longer expelled correctly: instead of flowing towards the aorta, part of it flows back towards the left atrium, in the opposite direction to normal circulation. This is known as mitral regurgitation.
Acutely, this leakage of blood to the left atrium and lungs can cause respiratory distress. In the long term, the heart, forced to compensate for this progressive insufficiency, gradually remodels until it is no longer able to maintain sufficient function: this is heart failure.
To date, no such leakage has been observed in astronauts’ hearts, mainly because this aspect has not yet been studied in detail. Further work is needed to assess the potential clinical implications of this finding in the space environment.
This new study raises as many questions as it answers. We don’t yet know whether papillary muscle atrophy is reversible after return to Earth, or whether it worsens on longer missions. We also don’t know whether it actually affects mitral valve function in the long term.
More specific imaging protocols, dedicated to valve assessment, and long-term follow-up will be needed to answer these questions, as mitral regurgitation can remain asymptomatic for years before irreversible damage occurs.
Space, a physiological "time machine
This discovery takes on its full meaning when placed in the context of terrestrial aging. On Earth, prolonged physical inactivity and physiological aging are associated with an increased risk of mitral insufficiency. This process takes place over years, even decades. In orbit, weightlessness compresses this time: within six months, similar anatomical changes appear.This is what makes space a physiological "time machine". Astronauts don’t actually age faster, but their bodies are subjected to stresses that reproduce some of the effects of aging in an accelerated and reversible way. This particularity offers researchers a unique window of opportunity to observe and understand mechanisms that, on Earth, are lost in the slowness of biological time.
The parallel between space and terrestrial aging extends far beyond the heart. The musculoskeletal system also undergoes rapid deconditioning in weightlessness: astronauts can lose up to 1-2% of their bone mass per month in certain skeletal regions, a rate ten times greater than that of terrestrial osteoporosis. Their leg muscles rapidly atrophy, unable to support the weight of their bodies.
Eyes are not left out either. In weightlessness, body fluids redistribute towards the head, increasing pressure within the skull and causing structural changes to the optic nerve and eyeball, a syndrome now well-documented as Spaceflight Associated Neuro-Ocular Syndrome (SANS).
Another study has shown that six months of spaceflight induces insulin resistance associated with stiffening of the arteries carrying blood to the brain. On Earth, this process takes years.
To combat this generalized deconditioning in space, the solution implemented on the ISS is intensive: astronauts follow a strict protocol of around two and a half hours of daily exercise, combining cycling, treadmill and muscle strengthening.
However, while this protocol considerably limits the loss of muscle mass, as well as total cardiac mass, it unfortunately does not prevent papillary muscle atrophy. This specific vulnerability could be explained by their unique anatomy and under-stimulation in a spatial context.
From orbit to hospital bed: a double benefit
The significance of these observations is twofold. For space agencies, they underline the need to monitor astronauts’ valve function, particularly with a view to long-term missions to the Moon or Mars. If papillary muscle atrophy worsens over time, it could theoretically compromise mitral valve tightness and lead to heart leaks in crews far from terrestrial medical assistance.They also have promising implications for "terrestrial" medicine: understanding how weightlessness causes papillary muscles to atrophy could help identify the mechanisms underlying their age-related or sedentary deterioration.
One thing is certain: the study of astronauts’ hearts continues to shed light on our understanding of terrestrial cardiovascular disease. Every space mission is also a medical mission that benefits the millions of heart patients on Earth. As we look to the stars, we learn to better care for those who have their feet firmly planted on the ground. Yet another paradox in the fascinating adventure of space exploration.
Cyril Tordeur , PhD Candidate in Biomedical Sciences and Pharmacy (Space Cardiovascular Physiology), Université Libre de Bruxelles (ULB ) This article is republished from The Conversation under a Creative Commons License. Read the original article.
